Cardiac arrest proves fatal for most patients. The rate of survival to hospital discharge after cardiac arrest is only 6.3%.[1]
Patients who had favorable neurological survival were even less at only 4.8%.1
This means for every twenty patients who suffer cardiac arrest, only one will survive with good or only moderate cerebral disability.1
When it comes to improving the odds of survival, the quality of CPR must be considered. High-quality CPR is associated with:[2]
- Higher rates of return of spontaneous circulation (ROSC)
- Improved cerebral perfusion
- Improved cerebral oxygenation
- Improved survival
- Improved neurological outcomes
Due to the impact CPR quality can have on patient outcomes, frequent assessment of CPR quality is important.2
Today’s article reviews current practices when it comes to CPR quality assessment and how regional oximetry (rSO2) can improve real-time CPR evaluation and clinical decision-making in the event of cardiac arrest.
CPR Quality Assessment Challenges
The American Heart Association Guidelines for CPR puts an emphasis on physiological monitoring to maintain quality CPR, but this remains a challenge for clinicians.[3][4]
Monitoring pulse, breathing patterns, and observing for consciousness are the default methods to evaluate the quality of CPR. Observational measures are used more often because physiological monitors available, including pulse oximetry and non-invasive blood pressure, tend to be unreliable during CPR administration.4
Other monitoring options like Intermittent ECG rhythm checks require chest compression interruption and end-tidal CO2 is only an option for intubated patients making these two methods impractical for all patients.4
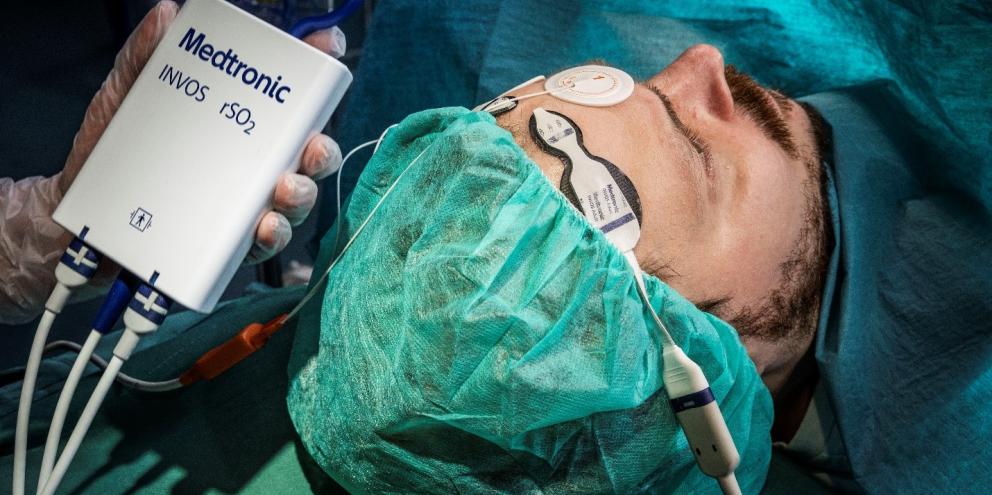
However, there is one physiological method that provides accurate information on brain oxygenation and perfusion without requiring interruption or intubation during CPR. Cerebral oximetry measures regional oxygen saturation and has been shown to be a reliable indicator of CPR quality and efficacy.4
Compared to pulse oximetry which needs a pulse to determine oxygenation – rSO2 measurements using near-infrared spectroscopy (NIRS) can be reliably taken during episodes of pulselessness.2 There is also no need to interrupt chest compressions.2
Challenges of Using Cerebral Oximetry During CPR
Although helpful, monitoring rSO2 during CPR has a few challenges clinicians must consider. Most challenges to rSO2 during CPR are related to the patient's condition rather than the CPR procedure itself.
Patient conditions can affect rSO2 values as well, including2 acid-base status, pulmonary function, drug use, skin pigmentation or extracranial contamination .
Ambient light can impact NIRS values making using rSO2 readings in outpatient settings, such as outside, a challenge.2
When deciding on intermittent versus continuous monitoring, research has shown intermittent measurements are not as helpful as continuous monitoring in evaluating CPR efficacy.2
Continuous, or dynamic, rSO2 monitoring rather than spot-checking provides information about cardiac output and cerebral perfusion.2
The use of regional oximetry to support clinicians in delivering better care and potentially improved outcomes
Even after successful resuscitation, there is a high risk of disability. The leading cause of disability after resuscitation is low cerebral perfusion causing a hypoxic reperfusion brain injury.2
By monitoring rSO2 with NIRS technology, clinicians can monitor oxygen metabolism during CPR and in turn, potentially improve patient outcomes.2
NIRS can monitor cerebral blood oxygenation during CPR as well as pulse rate in real-time.[5] Studies have shown when there is a lack of pulse rate indicated with NIRS, this can identify when chest compressions have become ineffective. This ability can help gauge the quality of CPR in real-time and assist clinicians with any adjustments.5
Adjustments including changing the person performing chest compressions have been shown to improve rSO2 further highlighting the benefit real-time quality monitoring can have on improving patient outcomes.[6]
Arrhythmias can coincide with changes in rSO2 measurements and potentially guide rapid clinical intervention. For example, episodes of ventricular fibrillation have been shown to coincide with decreases in rSO2 values.4 Declines in rSO2 values were also shown to predict re-arrest scenarios like pulseless arrhythmias or severe cerebral hypoperfusion.4 By predicting these events, CPR can be initiated or re-initiated more quickly.
Predicting Patient Outcomes
In addition to monitoring the quality of CPR, rSO2 monitoring during CPR may be able to help clinicians predict ROSC.2 A 2020 meta-analysis found patients with a high initial or mean rSO2 were associated with an increased incidence of ROSC.[7]
A 2015 review found in patients who had ROSC had much higher initial as well as average rSO2 values than those who did not. [8]
When looking at patients who survive at least one week after cardiac arrest, they have significantly higher rSO2 values during CPR.4
Another review found studies reported higher mean NIRS values in patients with ROSC, patients who survived to discharge, and patients who survived with good neurological outcomes.[9] The same review found if there was a failure to achieve at least a 30% NIRS saturation had a very low likelihood of ROSC.9
This type of information could help clinical decision-making regarding if or when to terminate resuscitation efforts as part of a multifactorial approach.9
In addition to predicting ROSC, rSO2 can help predict the risk of re-arrest events. Several studies have found decreases in NIRS values to be associated with re-arrest.2 This information can help clinicians be better prepared for patients they have identified as high risk of re-arrest.
One theory as to why NIRS can be a reliable predictor of ROSC is the ability to identify neurological injury and blood flow to the brain – especially since brain injury is a major determining factor of disability post-cardiac arrest.9
NIRS may help predict patient outcomes with its ability to determine the tissue oxygen index as well as any changes to the tissue oxygen index. Case studies have found patients who had a tissue oxygen index of 40% or greater had a significantly higher chance of ROSC after CPR.6
It’s worth noting it is difficult to determine a static cut-off value due to the amount of variability that may be present for each individual patient scenario. Clinicians still must use clinical judgment when interpreting rSO2 measurements and use them as a piece of the greater patient picture.9
GE HealthCare CARESCAPE monitors with INVOSTM Technology
GE HealthCare CARESCAPE monitors with INVOSTM regional oximetry provides seamlessly integrated rSO2 technology. With patented sensor spacing configurations and user-friendly design, INVOSTM technology is highly sensitive to oxygen saturation and blood flow changes.[10] This combined with an algorithm providing real-time actionable information during CPR, enables clinicians to optimize clinical decision-making.
Summary
- While helpful, CPR is rarely successful in preventing death or disability due to cardiac arrest
- CPR quality plays a role in cardiac arrest survival, but assessing CPR quality in real-time is challenging
- Regional oximetry using NIRS provides a real-time accurate assessment of CPR quality without the need for chest compression interruption and can monitor perfusion in the absence of a pulse
- rSO2 monitoring during CPR aids clinicians in decision-making and quality assessment and has the potential to improve patient outcomes after cardiac arrest
- GE HealthCare CARESCAPE monitors with INVOSTM regional oximetry provides integrated rSO2 monitoring solutions to monitor oxygen saturation and blood flow changes, even during CPR
The INVOS™ monitoring system should not be used as the sole basis for diagnosis or therapy and is intended only as an adjunct in patient assessment. INVOSTM is available on CARESCAPE monitors software version 3.2 onwards.
References
[1] Petek, B, Bennett D, Ngo, C, et al .(2019). Reexamination of the UN10 rule to discontinue resuscitation during in-hospital cardiac arrest. JAMA Network Open. 2(5):e194941.
[2] Takegawa, R et al. (2020). Near-infrared spectroscopy assessments of regional cerebral oxygen saturation for the prediction of clinical outcomes in patients with cardiac arrest: a review of clinical impact, evolution, and future directions. Frontiers in Medicine. 7: article 587930.
[3] Hamanaka, K et al. (2020). Cerebral oxygenation monitoring during resuscitation by emergency medical technicians: a prospective mulitcenter observational study. Acute Medicine and Surgery. 7(1): e528.
[4] Meex, I et al. (2013). Feasibility of absolute cerebral tissue oxygen saturation during cardiopulmonary resuscitation. Critical Care. 17: R36.
[5] Yagi, T et al. (2020). Usefulness of a new device to monitor cerebral blood oxygenation using NIRS during cardiopulmonary resuscitation in patients with cardiac arrest: a pilot study. Oxygen Transport to Tissue XLI. Advances in Experimental Medicine and Biology. Vol 1232. 323-329.
[6] Koyama, Y et al. (2013). A new method to detect cerebral blood flow waveform in synchrony with chest compression by near-infrared spectroscopy during CPR. The American Journal of Emergency Medicine. 31(10): 1504-1508.
[7] Liu, Y et al. (2020). Association between cerebral oximetry and return of spontaneous circulation following cardiac arrest: a systematic review and meta-analysis. PLOS One. 1-12.
[8] Sanfilippo, F et al. (2015. Cerebral oximetry and return of spontaneous circulation after cardiac arrest: a systematic review and meta-analysis. Resuscitation. 94:67-72.
[9] Cournoyer, A. et al. (2016). Near-infrared spectroscopy monitoring during cardiac arrest: a systematic review and meta-analysis. Academic Clinical Medicine. 23(8):851-862.
[10] Kim, M et al. (2000). Estimation of jugular venous O2 saturation from cerebral oximetry or arterial O2 saturation during isocapnic hypoxia. Journal of Clinical Monitoring Comput. 16(3):191-99.